Challenges in Diagnosing Infection in ED
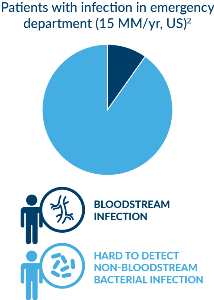
Acute infections account for 15 million visits to U.S. Emergency Departments every year. (1) Nearly all of these are challenging to the ED physician, who must consider the patient’s clinical presentation as well as the results of diagnostics studies that include imaging studies and various clinical tests to determine the patient’s diagnosis. Current diagnostic testing methods are largely inaccurate or too slow, resulting in delayed or inappropriate treatment. Not only does this harm patients but it furthers the growing and costly global problems of antibiotic resistance and sepsis.
What are the Challenges in Diagnosing Infections?
- Infections are diverse and no single test confirms or eliminates an infection. (2)
- A bacterial infection must be identified immediately so that it can be treated with antibiotics to prevent disease progression, including potential sepsis.
- Blood culture—the “gold standard” in diagnosing infections—misses at least 70% of bacterial infections and it is slow; (2) most infections never enter the bloodstream (e.g., pneumonia, skin, soft tissue).
- Most the testing approaches lead to negative results, even in patients with acute infections and as a result, are not actionable in the Emergency Department.
- Because of these diagnostic shortcomings, infections are frequently empirically—and incorrectly—treated with antibiotics, (3)which may lead to other problems. If the wrong antibiotic is prescribed, there is no benefit to the patient, and antibiotics can cause harmful side effects. Further, this practice contributes to antibiotic resistance. Conversely, bacterial infections which require antibiotics are sometimes missed.

Sobering Statistics: Antibiotic Resistance
- Each year in the U.S., at least 2 million people become infected with bacteria that are resistant to antibiotics and at least 23,000 people die as a direct result of these infections. (4)
- 30% of patients who get antibiotics don’t need them. (3)
- 80 million outpatient visits for suspected infections each year in the U.S. alone result in blind treatment of an antibiotic prescription. (3)
- Costs related to antibiotic resistance is expected to grow to over $100 trillion by 2050. (4)
Where does antibiotic resistance come from? Several mechanisms (5)can come into play to create “super bugs”. Selective pressure means that in the presence of an antimicrobial, microbes are either killed or, if they carry resistance genes, survive. These survivors will replicate, and their progeny will quickly become the dominant type throughout the microbial population. Another way resistance develops is during microbial replication-- mutations may arise and some of these mutations may help an individual microbe survive exposure to an antimicrobial, or gene transfer may occur. An area of debate is the agricultural use of antibiotics, which may increase resistance in microbes. When combined with the pressure that physicians are under from sick patients or patients who are insistent on receiving antibiotics, there is little surprise that the numbers of antibiotic resistant bacteria continue to climb.

Sobering Statistics: Sepsis
- Sepsis kills more than 250,000 people in the U.S. each year (6) and is estimated to cause or contribute to over 5 million deaths worldwide annually. (7)
- Sepsis accounts for almost half of all in-hospital deaths and is the leading cost for the U.S. healthcare system, at $27 billion each year. (8)
- There is current no single, definitive test for sepsis; rather, physicians typically rely on a battery of tests clinical criteria – all with sub-optimal performance. (9)
- Sepsis diagnosis requires two factors: presence of an infection, and a specific increase in organ dysfunction (as currently measured by an illness-severity scoring system known as SOFA—Sequential Organ Failure Assessment)
- Infections are difficult to diagnose (as mentioned) and SOFA in imprecise; current laboratory measures of organ dysfunction (e.g., procalcitonin, lactate) have poor specificity resulting in unnecessary hospital resource utilization.
Sepsis is a fast-progressing and life-threatening condition in which the body’s immune system, already fighting a severe infection, becomes dysregulated and damages its own tissues and organs. It is a medical emergency that requires prompt treatment.
New Ways of Diagnosing Acute Infections and Sepsis Are Coming: TriVerity™ acute infection & sepsis test
The TriVerity acute infection & sepsis test is a rapid (<30 minutes) point-of-care diagnostic for use in emergency medicine and critical care. The test is uniquely effective because it
- Diagnoses acute infection or sepsis even when pathogens have not entered the bloodstream or aren’t detectable – the majority of infected patients – by assessing the patient’s immune (host) response (10)
- Addresses what matters most for rapid treatment decisions
- Presence of infection
- Severity
- Assists in patient management decision and antimicrobial stewardship by helping answer:
- Does the patient require antibiotics, or rest and a flu treatment?
- What level of care does the patient need?

Interested in being an early evaluator of the TriVerity test?
Send us an email at info@inflammatix.com with your name and contact information so we can notify you. Or, for more information visit us at www.inflammatix.com.
References
- Horeczko, T, et al. Epidemiology of the Systemic Inflammatory Response Syndrome (SIRS) in the emergency department. The Western Journal of Emergency Medicine. 2014 May;15(3):329-336. DOI: 10.5811/westjem.2013.9.18064.
- Coburn, B, et al. Does this adult patient with suspected bacteremia require blood cultures? JAMA. 2012;308(5):502-511.
- Fleming-Dutra, KE, et al. Prevalence of inappropriate antibiotic prescriptions among US ambulatory care visits, 2010-2011. JAMA. 2016;315(17): 1864-1873.
- Donnelly, JP, et al. Antibiotic utilization for acute respiratory tract infections in U.S. emergency departments. Antimicrob. Agents Chemother. 2014; (3):1451-7. Epub 2013 Dec 16.
- https://www.niaid.nih.gov/research/antimicrobial-resistance-causes
- The Rory Staunton Foundation for Sepsis Prevention website (https://rorystauntonfoundationforsepsis.org/what-is-sepsis/)
- Fleischmann C, et al. Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations. Am J Respir Crit Care Med. 2015; 193: 259e72.
- Statistical Brief #225. Healthcare Cost and Utilization Project (HCUP). June 2017. Agency for Healthcare Research and Quality, Rockville, MD. www.hcup-us.ahrq.gov/reports /statbriefs/sb255-inpatient-US-Stays-Trends.jsp.
- Singer, M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801—810.
- Gunsolus, IL, et al. Diagnosis and managing sepsis by probing the host response to infection: advances, opportunities, and challenges. JCM. DOI: 10.1128/JCM.00425-19.

