Intracranial Hemorrhage
Original Author: Zoe Oliver, MD, CCFP(EM), University of Manitoba, Rady Faculty of Health Sciences
Revisions Author: Holly Caretta-Weyer, MD, MHPE(c), Stanford University
Editor: Chaiya Laoteppitaks, MD, Sidney Kimmel Medical College,
Last Update: November 2019
Case Study
Caitlin is a 37 y.o. female who presents to your Emergency Department via EMS. She was running on a treadmill when she had the sudden onset of a “thunderclap” headache. EMS notes that upon their arrival she was seated with her head in her hands. She was complaining of extreme photophobia and a severe headache. She has become more lethargic during transport.
The patient arrives in the room and is observed to be writhing in pain with her eyes closed. She is able to verbalize that her head hurts but cannot answer specific questions about the events leading to her arrival in the Emergency Department. Vital signs are: T 37C, P 110, BP 150/100, RR 20, O2 98%.
Airway - is intact.
Breathing - she is tachypneic but lungs are clear.
Circulation – she is tachycardic and hypertensive.
Disability – she is oriented to self and place but will not open her eyes, is able to follow only simple commands and will respond appropriately to only some questions, and moves all extremities to pain. GCS 12 (E1, V5, M6).
Exposure – no evidence of traumatic injuries.
Objectives
Upon completion of this self-study module, the student should be able to:
1. List the first steps in managing a patient with a deteriorating level of consciousness.
2. Describe the classical history and physical exam findings associated found with intracranial hemorrhage (ICH)
3. Identify different types of hemorrhage seen on computed tomography (CT) images of the brain
4. Interpret lumbar puncture results in the assessment of possible subarachnoid hemorrhage (SAH)
5. Summarize the first steps in managing a patient with an intracranial hemorrhage
Introduction
Bleeding within the fixed vault of the cranium is a life-threatening emergency that affects around 65,000 patients a year in the United States of America. Intracranial hemorrhage can cause increased intracranial pressure, which in turn damages the brain parenchyma and can cause herniation. This can lead to permanent neurologic deficits or death. Rapid diagnosis and intervention by the emergency physician can be a crucial determinant of patient outcomes.
Initial Actions and Primary Survey
A patient with any type of intracranial hemorrhage may present with coma, rapidly declining level of consciousness, or seizure. In such cases, the priority is the ABCD’s.
Secure the Airway if there are concerns about oxygenation, ventilation, airway protection, prolonged seizure, or rapidly deteriorating clinical status. A neuroprotective rapid-sequence intubation protocol is preferred.
Proceed with a brief assessment of Breathing, Circulation, and Disability while the patient is being pre-oxygenated, and intubation equipment and drugs are prepared. Neurosurgeons often find the documentation of a pre-intubation neurological exam to be helpful in determining prognosis. At minimum, such an assessment should include documentation of the Glasgow Coma Score, the pupillary size and reactivity, and motor strength in the four limbs. Sensation and reflexes can be included if time permits.
Make sure to check a fingerstick glucose before intubating.
It is important to be able to recognize Cushing’s triad, as this is a sign of impending brain herniation. Cushing’s triad describes the physiologic response to rapidly increasing intracranial pressure (ICP) and imminent brain herniation. Its features are:
- Hypertension
- Bradycardia
- Abnormal respiratory patterns
Other signs of imminent herniation are lack of pupillary reaction and/or pupillary asymmetry. Signs of imminent herniation necessitate emergent intervention (see ‘Treatment’).
Presentation: Four categories of ICH
Intracranial hemorrhages (ICH) fall into four broad categories:
- Epidural hematoma
- Subdural hematoma
- Subarachnoid hemorrhage
- Intracerebral hemorrhage
It is important to understand the difference between the terms intracranial hemorrhage and intracerebral hemorrhage. The former refers to all bleeding occurring within the skull, while the latter indicates bleeding within the brain parenchyma.
All intracranial hemorrhages (ICH) share some classic clinical features. Common presenting symptoms include headache, nausea, vomiting, confusion, somnolence, or seizure. There is a wide clinical spectrum: patients may be alert and conversant, or moribund. In elderly, alcoholic, and anticoagulated patients, even minor head trauma can result in devastating intracranial bleeding.
Despite these commonalities, there can be differences in the presentation of the four types of ICH, discussed by category below.
Subarachnoid Hemorrhage
The classic presenting symptom of SAH is an acute onset “thunderclap” headache that may be accompanied by loss of consciousness, vomiting, neck stiffness, or seizure. Thunderclap headaches reach maximum intensity within seconds. The headache is often occipital in location. A significant proportion of patients (30-50%) will also have a warning (sentinel) headache – this is a small bleed which heralds a much larger, potentially catastrophic event.
The Hunt and Hess Grading System( Table. 1) is one method used to classify the severity of illness in patients with SAH.
Table 1. Hunt and Hess Grading System for SAH
Recent exertion, hypertension, excessive alcohol consumption, sympathomimetic use, and cigarette smoking are risk factors for both SAH and intracerebral bleeds. However, the strongest risk factor for SAH is family history, which carries a 3- to 5-fold increased risk.
Most SAH are due to the rupture of saccular aneurysms. It is important to note that many in the general population have aneurysms that are asymptomatic, and will not rupture.
Epidural Hematomas (EDH)
Epidural hematomas are accumulations of blood between the skull and the dura, and typically occur after significant blunt head trauma. Fractures of the temporal bone can disrupt the middle meningeal artery, leading to high-pressure bleeding within the cranial vault. Herniation can occur within hours if the hematoma is not evacuated, so early recognition is key.
The classic description of an EDH is a brief loss of consciousness after a blow to the head, followed by a lucid period. Soon after, the level of consciousness deteriorates again, possibly progressing into herniation and death. You might hear this described as the ‘talk and die’ phenomenon. In reality, most EDH patients either do not lose consciousness or do not regain it once lost.
Subdural Hematomas (SDH)
Subdural hematomas are extra-axial blood collections between the dura and the arachnoid mater. Subdural hematomas form when bridging veins are sheared during acceleration-deceleration of the head.
Since the bleeding is venous and low-pressure, the hematoma can grow fairly slowly and the presentation can be delayed by days to weeks. This is particularly true in patients with brain atrophy, whose bridging vessels are more susceptible to shear and who can more readily accommodate the additional intracranial blood volume. These patients can present with an indolent decline in mental status, confusion, fatigue, or other subtle changes.
Subdural hematomas have a wide clinical spectrum. Rapid accumulation of extra-axial blood, the absence of pre-existing atrophy, and the presence of other traumatic brain injuries correspond to a worse neurologic status at presentation. As the younger brain is less atrophic, even small volumes of extra-axial blood can increase ICP and result in severe deficits.
In the pediatric population, presence of acute or chronic SDH should raise suspicion of for child abuse (non-accidental trauma), although SDH can also occur as a result of birth trauma. The physician should search for other signs of ‘Shaken Baby Syndrome’, including retinal hemorrhages and long bone fractures. Infants with increased ICP might present with a bulging fontanelle, enlarged head circumference, emesis, failure to thrive, and seizures.
Chronic SDH is a grand imitator. It occurs more often in elderly and alcoholic patients as they are most prone to atrophy and/or coagulopathy. The most common presentation is altered mental state. Hemiparesis, headache, and falls are other possible features. As the symptoms can be subtle, the differential diagnosis broadly encompasses any potential cause of weakness or confusion in the elderly. This highlights the need to have a low threshold for CT scanning the head of any elderly patient with a change in mentation that is not convincingly explained by other pathologies.
Intracerebral Hemorrhage
Intracerebral hemorrhage(ICH) is bleeding within the parenchyma of the brain. This can be secondary to numerous etiologies including uncontrolled hypertension, ruptured saccular aneurysms, vascular malformations or traumatic injury. Hypertensive vasculopathy is the most common etiology of spontaneous ICH. Elevated blood pressure causes damage to the small vessels in the brain, weakening the vascular wall and making them more prone to rupture. These changes are seen in those with chronically elevated blood pressure. ICH can occur during intense activity or emotional upset, however most occur during regular activities.
Symptoms of acute ICH are going to be similar to other etiologies of head bleed. Typically patients will present with headache, nausea, vomiting and depressed mental state. Patients may also present in a comatose state or severely obtunded. Rapid management of these patients is critical to avoid further clinical decline. Presenting symptoms are going to be related to the area that is affected by the ICH. Rapid diagnosis and aggressive treatment of blood pressure is crucial to prevent further extension of areas of clot. Reversal of anticoagulation is also crucial in early management.
Diagnostic Testing
CT Scan
The head CT is the mainstay of diagnosis in ICH. On CT, acute bleeding appears hyperdense (whiter) relative to the surrounding tissues. The subacute phase occurs between days 3 and 14, when blood becomes isodense to the brain parenchyma. It is particularly easy to miss intracranial bleeding during this phase as the blood and the brain may appear the same shade of grey. After about two weeks, blood appears hypodense (darker) relative to the brain.
The standard diagnostic pathway for SAH used to include CT followed by LP. This was because the sensitivity of CT, while reasonable, was still in the low 90’s and missing a diagnosis of SAH is potentially lethal. So many sources used to advocate that an LP was necessary in all cases of suspected SAH in order to conclusively exclude the presence of blood in the CSF.
However, improvements in CT scanning technology have led to improved sensitivities for SAH, particularly within the first 6 hours (99-100%). As such, the majority of clinicians have moved away from the ‘mandatory LP’ if the patient’s CT is normal within the first 6 hours after onset of symptoms. It is important to note that sensitivity can be affected by both the generation of the scanner and the experience of the reader.
Large volumes of blood in the cranium can cause radiologic signs of increased intracranial pressure. These include:
- Midline shift
- Ipsilateral compression of the ventricles with or without contralateral ventricular enlargement
- Obliteration of the sulci
- Blurring of the grey-white junction
- Subarachnoid Hemorrhage
A subarachnoid hemorrhage appears as blood in the ventricles, sulci, and cisterns on CT. An example of a CT scan showing SAH is shown in Figure 1. The overall sensitivity of CT for subarachnoid hemorrhage is best within the first 6 hours. Sensitivity declines with time, particularly with detection of smaller bleeds.
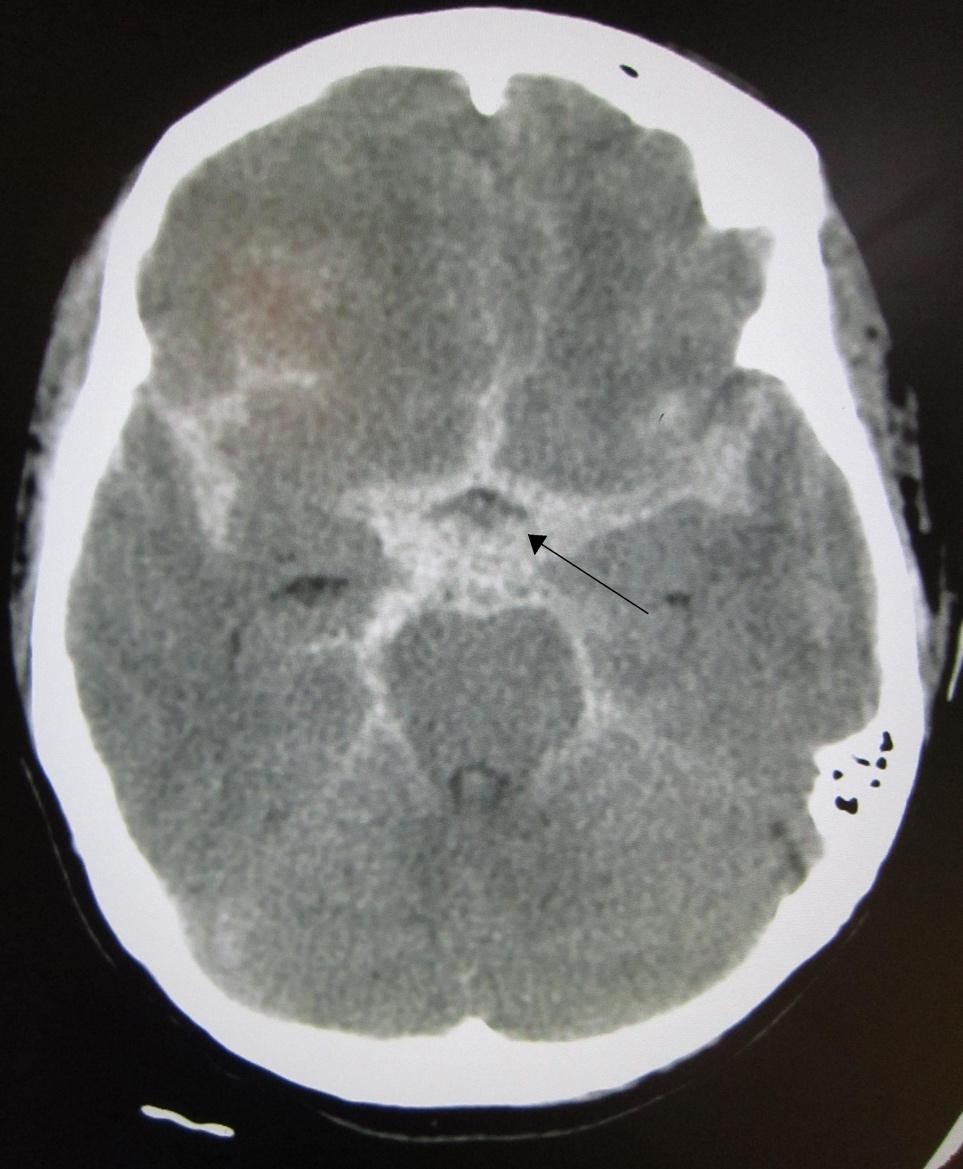
Image 1. CT Scan showing Subarachnoid Hemorrhage (SAH). Arrow added demonstrating area of SAH. Image courtesy of : https://commons.wikimedia.org/wiki/File:SAH1.JPG. This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
Subdural Hematoma
Subdural hematomas appear convex, or crescent-shaped, on CT and may cross suture lines. An example of a CT scan of a subdural hematoma is shown in Image 2.
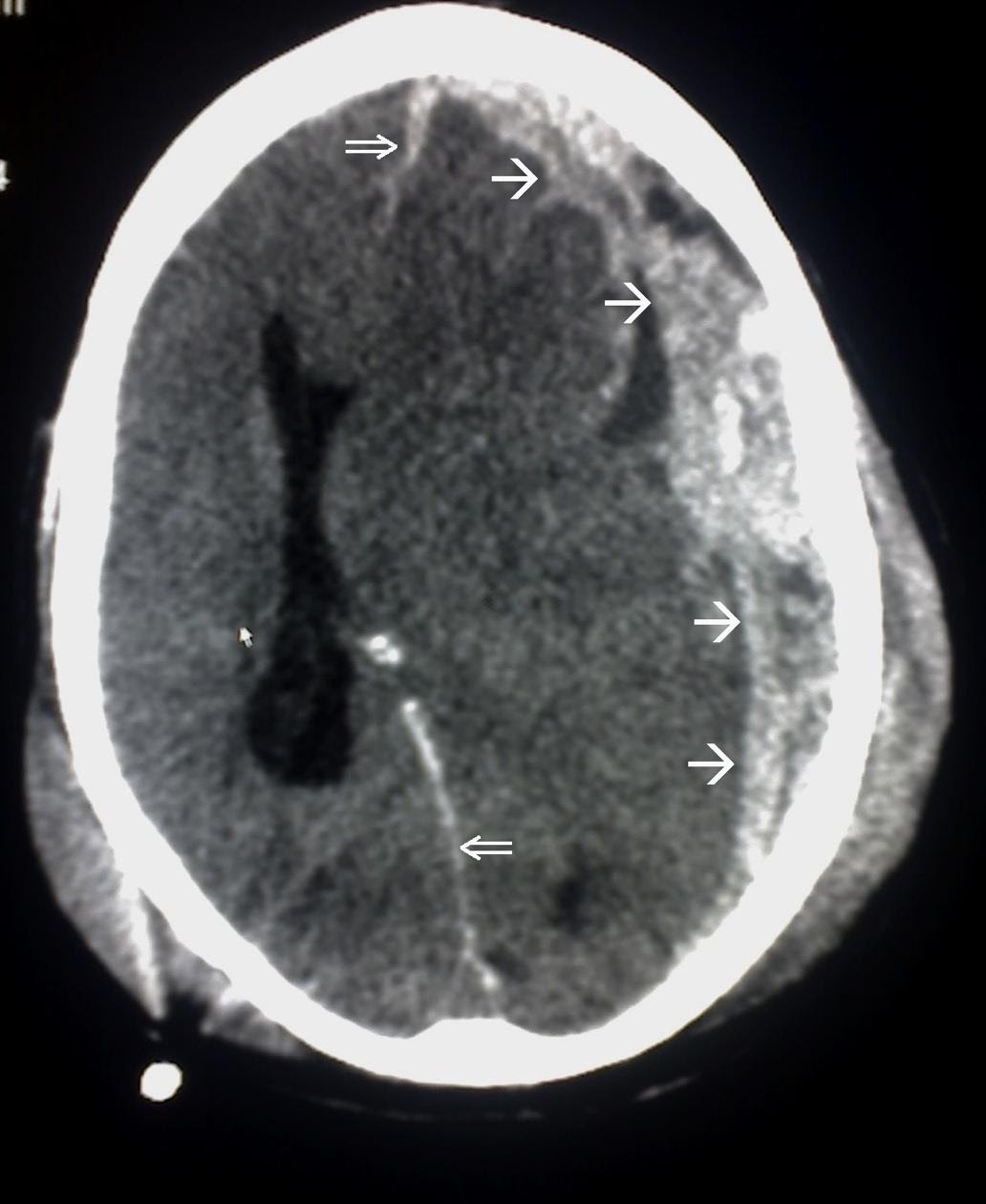
Image 2. Subdural Hematoma on CT scan. Border of the SDH is denoted by the white arrows ( added for emphasis) Image courtesy of : https://commons.wikimedia.org/wiki/File:Trauma_subdural_arrows.jpg This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
Epidural Hematoma
Epidural hematomas appear convex, or lens-shaped, on CT and do not cross suture lines. An example of a CT scan of an epidural hematoma is shown on Image 3.
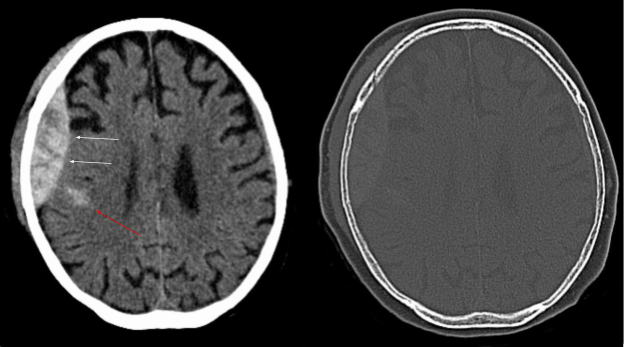
Image 3. CT scan of an epidural hematoma (white arrows). Red arrow demonstrates an area of intracerebral hemorrhage. Arrows have been added Image courtesy of: https://commons.wikimedia.org/wiki/File:Epidurale_Blutung_-_CT_-_WT-KF.jpg, This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
Lumbar Puncture
In cases of suspected SAH with a negative CT, lumbar puncture is often the second diagnostic step, particularly if the CT is delayed more than 6 hours after the onset of symptoms. An excellent (but password protected) tutorial video on lumbar puncture is available through the New England Journal of Medicine website (try accessing through your university library) – referenced below. Ultrasound is now being used more frequently to identify the landmarks for the LP; for a video on the technique follow the link in the references.
Two CSF features are most important:
- Absence or clearing of blood.
- Xanthochromia
Normal CSF does not contain red blood cells. Blood in the CSF may be a result of SAH, infection, or a traumatic tap. Many sources state that if the number of red blood cells decreases by 50% from tube 1 to tube 4, the blood can be attributed to a traumatic tap. However, this decrease can occur in SAH as well, so the tap should only be labeled ‘traumatic‘ if the fourth tube is almost completely free of blood (less than 5 RBC’s per high powered field).
Xanthochromia refers to a yellow or pink discoloration of the supernatant once the CSF is centrifuged. It results from the breakdown of blood cells within the CSF. Xanthochromia is determined with either visual inspection or spectrophotometry; the latter is less commonly available. The presence of xanthochromia is highly specific for SAH.
If the CT or LP results are consistent with SAH, some form of angiography is necessary. While conventional digital subtraction angiography (DSA) is the gold standard, it may be less readily available than CT or MR angiography. Since CT angiography is rapid and non-invasive, it is commonly used to identify saccular aneurysms once the diagnosis of SAH is confirmed.
How do I make the diagnosis?
- Suspect the illness. Complete a good history and physical exam.
- Order a CT head when appropriate. When there is a traumatic mechanism, you might consider using the Canadian CT Head Rules.
- Know the limitations of CT: small SAH’s or those with delayed presentation might be hard to spot on CT.
- Subdural and epidural hematomas can also be subtle.
Please see the below link for further information regarding the Canadian CT head rules and an interactive check-list,. The rules can be used if all criteria of the rules are met.
https://www.mdcalc.com/canadian-ct-head-injury-trauma-rule
Treatment
Medical Treatment
Some tenets apply to all patients with intracranial hemorrhage:
- Assess and frequently reassess the ABCD’s
- Discontinue or reverse anticoagulation
- Prevent hypotension and hypoxemia
- Control ICP, which often requires lowering the systemic blood pressure to a target systolic blood pressure less than 140 mm Hg
- Prevent seizure: prophylaxis may be necessary depending on the type and extent of bleeding
- Treat fever and infection aggressively
- Control blood glucose (target 140-185 mg/dL)
ICP control can be managed by:
- Monitoring/lowering blood pressure in consultation with Neurosurgery
- Elevating the head of the bead to 30 degrees
- Providing adequate sedation and analgesia
- Providing antiemetics as needed as vomiting increased intracranial pressure
- If signs of rapidly rising ICP or herniation, considering mannitol, hypertonic saline or mild hyperventilation (target CO2 around 30 mmHg)
Surgical Treatment
In 2006, a comprehensive set of guidelines for the surgical management of intracranial emergencies was published in the journal Neurosurgery. As a medical student, your focus should be on facilitating timely consultation with Neurosurgery for all patients with intracranial hemorrhage, unless it is clear that surgical or intensive care management would be against the patient’s wishes.
Disposition
The vast majority of patients with intracranial hemorrhage require close observation in either an intensive care unit or neurosurgical ward. Most will require intensive physiotherapy and occupational therapy before hospital discharge.
Pearls and Pitfalls
Obtain a very careful history from the patient regarding their headache, including any head injuries, use of blood thinners, onset and time course of the headache, associated neurological symptoms, and any other risk factors for intracranial hemorrhage.
In addition to a thorough neurological examination, make sure to examine the patient’s neck as well.
If indicated, ensure that you order the non-contrast head CT early! The sensitivity deteriorates significantly over time.
Lumbar puncture is still an important part of the management in patients whose CT scans are delayed more than 6 hours after the onset of the headache.
If your patient consents to an LP, be sure to warn them of the risk of post-LP headache (frequency ranges 10-20%) and to instruct them to lie flat for at least 30 minutes after the procedure.
Case Study
The patient’s GCS rapidly deteriorates, and she is intubated for airway protection. She is taken to CT, where her non-contrast head CT reveals a subarachnoid hemorrhage. She is started on nicardipine infusion to manage her blood pressure and hypertonic saline to lower her intracranial pressure. Additionally, the team elevates the head of the bed to 30 degrees. Neurosurgery is consulted. They place an intraventricular device to monitor her intracranial pressure and admit her to the neurosurgical ICU.
References
- 3D How To: Lumbar Puncture Procedure. SonoSite. https://www.sonosite.com/media-library/3d-how-lumbar-puncture-procedure.
- Blok KM, et al. CT within 6 hours of headache onset to rule out subarachnoid hemorrhage in non-academic hospitals. Neurology. 2015;84(19):1927-32.
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of acute epidural hematomas. Neurosurgery 2006; 58:S7.
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of acute subdural hematomas. Neurosurgery 2006; 58:S16.
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/american Stroke Association. Stroke 2012; 43:1711.
- Ellenby MS, et al. Lumbar puncture. N Engl J Med. 2006;355:e12. https://www.nejm.org/doi/full/10.1056/NEJMvcm054952.
- Epidural hematoma. https://commons.wikimedia.org/wiki/File:Epidurale_Blutung_-_CT_-_WT-KF.jpg.
- Marx J, Hockberger R, Walls R. Rosens Emergency Medicine Concepts and Clinical Practice. Philadelphia: Mosby/Elsevier; 2013Perry JJ, Stiell IG, Sivilotti ML, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: prospective cohort study. BMJ 2011; 343:d4277.
- Subarachnoid hemorrhage. https://commons.wikimedia.org/wiki/File:SAH1.JPG.
- Subdural hematoma. https://commons.wikimedia.org/wiki/File:Trauma_subdural_arrows.jpg.
Selected Online Resources
- Stroke Center: General information and neuroradiology files
- Ottawa Research Institute: Information regarding Canadian CT Head Rules
- University of Hawaii Pediatric Radiology: Extensive teaching file on pediatric ICH
- University of Iowa Radiology: Cerebrovascular radiology teaching file
