Rhythm Interpretation
Objectives
Upon finishing this module, the student will be able to:
- Understand the basic concepts of pediatric electrocardiogram (ECG) interpretation.
- Differentiate pediatric ECG findings from those seen in adults.
- Describe the pathophysiology that leads to pediatric ECG findings.
- Describe arrhythmias and cardiac anomalies often seen in the pediatric population.
Contributors
Update Author: Kendra Fabian, MD.
Original Authors: David Tillman, MD; Vanessa Tamas, MD; and Jamie Hess, MD.
Update Editor: Lisa Armstrong, MD.
Original Editor: S. Margaret Paik, MD.
Last Updated: November 2024
Introduction
A stepwise approach in the evaluation and interpretation of an electrocardiogram (ECG) is critical. Comprehensive instructions on a systematic approach to analyzing ECGs are beyond the scope of this chapter, but such an approach maximizes sensitivity and specificity. In general we recommend approaching all ECGs with the following ordered steps:
- Patient name/ID and age
- Rate
- Rhythm
- Waves and complexes: P, QRS, T
- Intervals: PR, QRS, QT
Utilizing the same systematic approach in analyzing pediatric ECGs that you employ with adults will ensure that you have a thorough understanding of the cardiac physiology of your patient. A consistent and stepwise approach to this facilitates a more ready appreciation of pathology and/or atypical ECG patterns, especially when seen in the context of the associated clinical presentation.
Key Differences in Pediatric ECGs
In contrast to adult ECGs, pediatric ECGs exhibit several key differences. Fortunately, the majority of these differences are predictable based on the development and changing physiology of a growing child. Given the significant physiologic changes that occur in the transition from prenatal to postnatal circulation, neonatal ECG is perhaps the most distinctly different, and thus potentially challenging to interpret when compared to an adult ECG.
In utero, pulmonary vascular resistance is high and systemic vascular resistance is low, resulting in a dominant right ventricle from the neonatal period through early toddler-age. As infants and children age, they concurrently increase their mass and cardiac output, and the structure and physiology of the pediatric ECG begins to more closely resemble an adult ECG. By late adolescence, the pediatric ECG can generally be interpreted with the same lens as an adult ECG. Patient age directly affects expected norms of ECG parameters, specifically as it relates to rate, intervals, and voltage. Thus, it is essential to know, or have ready access to, the age-adjusted ECG normal parameters for rate, axis, and intervals.
Normal ECG Parameters by Age
| HR (bpm) | QRS axis (°) | PR Interval (ms) (upper limit for age) | QRS Duration (ms) (upper limit for age) | |
| Neonate | 90-180 | 30-180 | 120 | 70 |
| Infant | 110-160 | 5-125 | 140 | 75 |
| 1-5 years | 100-150 | 5-110 | 170 | 75 |
| 6-12 years | 80-120 | 20-120 | 190 | 85 |
| 12+ years | 60-100 | -30-105 | 210 | 100 |
The Neonatal Heart
Due to their smaller muscular size, electrical impulses travel more quickly, leading to shortened durations and intervals. Additionally, the heart rate is increased. Electrical durations and intervals seen on ECG will increase and the heart rate will decrease as the patient ages and the heart enlarges.
- Heart Rate: neonatal heart rates are faster as the small, less-compliant heart is dependent on heart rate rather than stroke volume to maintain cardiac output. The normal rate decreases from 140 bpm at one month of age to 120 bpm by two years of age, to 90 bpm by ten years of age, and to 60-70 bpm for adolescents (approaching adult norms).
- QT Interval: The QT interval is a reflection of the function of ion channels involved in repolarization, and not myocardial thickness; it is most strongly modified by the HR. The baseline elevated pediatric heart rate increases the corrected QT interval (QTc). QTc = QTmeasured/HR.
- Voltages: Neonatal voltages are overall smaller, and scale (the number of millimeters used to represent one mV) is often changed to optimize the ECG display.
- Impulses: The timing of electrical impulses is faster in the smaller heart. The PR interval is shorter in the neonate (<120ms) and slowly increases to the adult level of ~160ms by late adolescence. Similarly, the QRS duration is shorter in the neonate (<80ms) and lengthens to the adult range of ~90ms as the heart enlarges.
Dominant Right Ventricle
The left ventricle (LV) in the adult is larger than the right ventricle (RV), and thus accounts for the majority of electrical forces. In the pediatric patient, the RV is relatively larger than the LV. Due to the relatively increased stress placed on the RV in utero (by pumping blood through the higher-resistance pulmonary circulation), it is the dominant ventricle at birth. The ECG during the neonatal and early childhood period will therefore show signs of RV dominance.
- Right Axis Deviation: In the very young child (up to one-two months), there is rightward deviation (greater than +90 degrees) to the QRS axis in the frontal plane. This normalizes to around +60 degrees by approximately six months of age.
- Juvenile T-Waves: T-waves are normally upright in lead V1 during the first week of life, but T-waves in leads V1-V3 are frequently negative or inverted in an infant or young child. T-waves in V2 and V3 usually become upright by adolescence. T-waves are not normally inverted in V5 or V6. Many children will retain their inverted T-wave in V1 into adulthood.
- Pseudo RBBB Pattern: The neonatal RV has a greater muscular mass (and therefore a greater electrical presence on ECG) than the adult RV. This leads to a pseudo-right bundle branch block (RBBB) pattern (RSR' in the right precordial leads [V1-V3] with dominant R-waves in the right precordium and dominant S-waves in the left precordium).

Figure: Many of the typical features of pediatric ECG can be seen here, including T-wave inversion V1-V3, dominant R-waves in V1-V3, and RSR' in V1. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
Vanessa is a nine-month-old girl with a history of a full-term gestation, without perinatal complications, presenting to the emergency department with her parents. They report that she has generally been feeding great, growing and developing well, but after daycare today seems fussy and a little sweaty. They deny any fever, and daycare told the parents that she didn't eat well during the day, and did not want to take her evening bottle, which is very unusual for her. Initial vitals are: heart rate 282, respiratory rate 30, SpO2 97%, blood pressure 94/62, and temp 37.6C.
On exam, she is alert but fussy. She has moist mucous membranes. She is tachypneic but without any abnormal lung sounds or increased work of breathing. You don't hear a cardiac murmur but her hart rate is so fast that it's difficult to be sure. Her abdomen is soft, non-tender, and she has good distal pulses in bilateral lower extremities. You obtain the following electrocardiogram after seeing her vital signs.
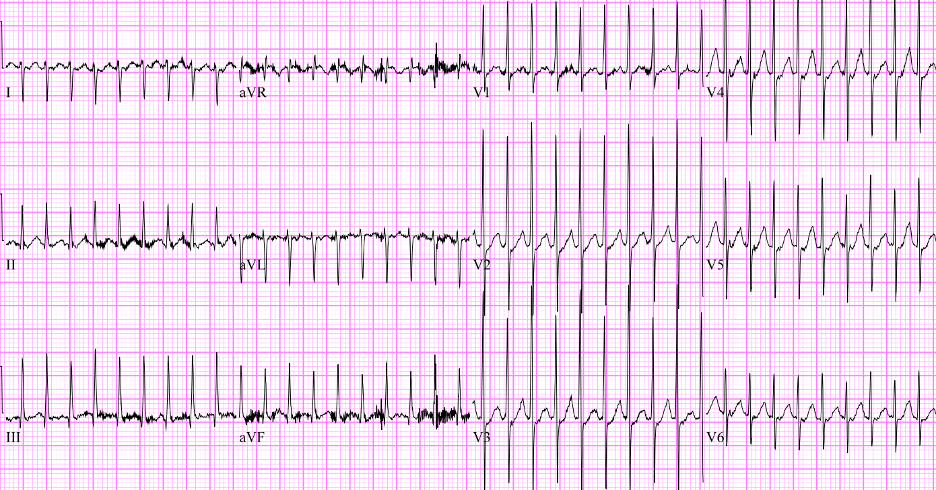
Figure: EKG of patient. Image courtesy of EM Sim Cases. Image used under the Creative Commons Attribution 4.0 International License.
Arrhythmias
Supraventricular Tachycardia (SVT)
In pediatric patients, SVT is the most common tachydysrhythmia. Most causes of SVT are AV reentry tachycardias (a recurrent cycle of electrical activity), which are distinct from ectopic atrial tachycardias (an abnormally fast-firing source of electricity). SVT is recognized by narrow QRS, regular RR interval, absent or difficult to define P-wave activity, with an elevated heart rate. As infants have an elevated baseline HR compared to adults, in SVT their rate may be between 220-320 bpm. Older children in SVT will typically have HR between 150-250 bpm.
Wolf Parkinson White (WPW) is an accessory pathway that conducts electricity between the atria and the ventricles. It can lead to pre-excitation (early partial depolarization of the ventricles), which manifests on an ECG as a slurred upslope at the beginning of the QRS complex ("delta wave"), causing a shortened PR interval. Keep in mind, the baseline shorter PR interval criteria in WPW are different in adults and children (the adult PR interval lower limit of normal is 120 ms, vs. 75 ms for an infant). The accessory pathway in WPW may not become clinically apparent until the child presents with SVT. There are two subtypes of SVT in WPW:
- Orthodromic ("correct running"), wherein the signal travels down the AV node to the ventricles via the normal rapid conduction pathway, causing a narrow QRS. Then, the electrical signal will travel up to the accessory/WPW pathway to re-excite the atria. This is the predominant pathway.
- Antidromic ("incorrect running"), wherein the electrical signal travels down the WPW pathway, distributes via myocyte-myocyte conduction, and causes a slow ventricular depolarization (i.e. wide QRS). The electrical signal then travels back to the AV node to re-excite the atria.
SVT can be treated with vagal maneuvers (e.g. dive reflex, the application of ice packs to the face blowing through a straw) or pharmacologic or electrical cardioversion. If SVT is refractory to vagal maneuvers, pharmacologic cardioversion with adenosine (0.1 mg/kg) is used. If the first attempt is unsuccessful, a second attempt at double the dose (0.2 mg/kg) can be tried. If the patient has evidence of hemodynamic instability, electrical synchronized cardioversion is used. SVT in WPW is treated the same as SVT in a normal heart (vagal maneuvers, adenosine, synchronized cardioversion). Verapamil (calcium channel blocker) should be avoided in infants less than one year of age as it can cause accessory pathway acceleration in this age group.
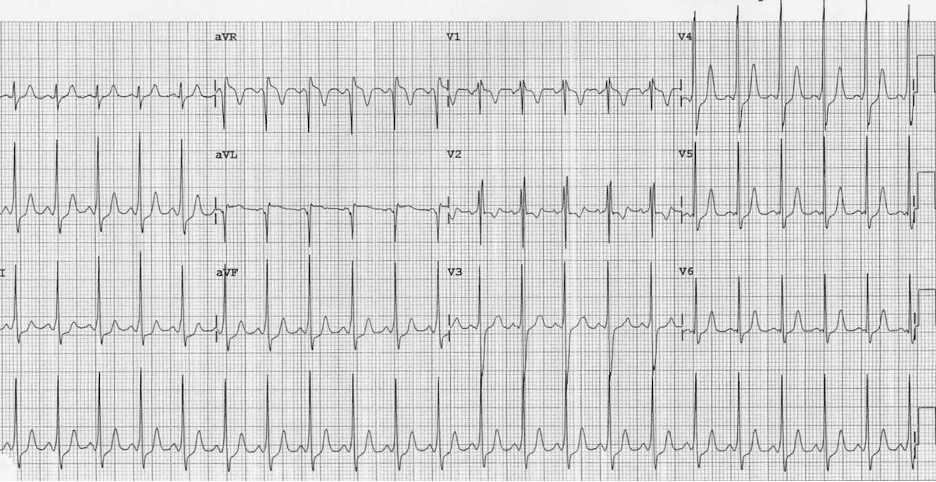
Figure: WPW in five-year-old male. Please note the shortened PR interval and slurred upstroke of QRS complex. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
Heart Block (AV Block)
Atrioventricular (AV) block is a disturbance in the conduction of electrical impulses from the atria to the ventricles. The same categories and treatment strategies that are applied in the adult population hold true in the pediatric population.
- First degree heart block: The PR interval is prolonged beyond the age-appropriate level, but there are no 'dropped beats.' This is often asymptomatic and requires no treatment.
- Second degree heart block: This is broken down into "Mobitz I (Wenckebach)" and "Mobitz II." Mobitz I is characterized by progressive prolongation of the PR interval, resulting in dropped beats. This can be seen following cardiac surgery, specifically in children after a congenital heart defect repair. Mobitz II is characterized by a fixed PR (quasi-normal or prolonged) with frequent dropped beats (often in a 3:1 or 4:1, conducted:dropped pattern). This can devolve into complete heart block and warrants cardiology evaluation for potential pacemaker placement.
- Third degree/complete AV block: The atrial rhythm and the ventricular rhythm are completely dissociated. The PP interval is often regular but does not correlate to the RR interval, which is often also regular. P-waves are seen in every portion of the cardiac cycle (e.g. sometimes buried in the T-waves, or in the QRS complex, or at varying places between the T and QRS). The QRS morphology is dependent upon the location of the ventricular pacemaker. For example, if the signal to fire the ventricles comes from the Bundle of His, the QRS will be narrow. However, if it arises from the left ventricle, the depolarization will travel more slowly to the right ventricle, leading to a wide QRS with a RBBB pattern. Third degree AV block requires evaluation by cardiology/electrophysiology for pacemaker placement.
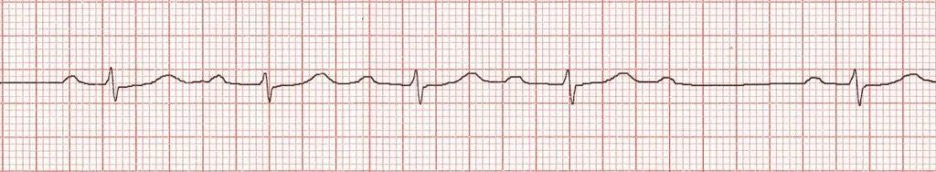
Figure: Second degree heart block, Mobitz I. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
There are certain conditions in the pediatric population that can be identified on electrocardiogram. Abnormal pediatric ECG findings can reflect acute or chronic infectious or inflammatory processes, underlying congenital arrhythmia or congenital cardiac defects. Below are some of the more common pediatric cardiac pathology and their associated ECG findings.
Anomalous Left Coronary Artery Originating from the Pulmonary Artery (ALCAPA)
This describes a congenital cardiac abnormality in which the origin of the left coronary artery (LCA) is in the pulmonary artery and not the aorta. This causes myocardial ischemia in the territory of the LCA and can manifest after birth with signs of heart failure. Classically, prominent Q-waves in the lateral leads (I, aVL, V3-V6) are seen on ECG. Diffuse ST-T segment elevation may be seen in neonates with this condition.
Hypertrophic Cardiomyopathy (HCM)
This is a condition in which some of the heart is enlarged, usually the interventricular septum with or without some hypertrophy of the left ventricle. The hypertrophy of the left ventricle can cause arrhythmias, both from architectural distortion as well as from dynamic obstruction of the left ventricular outflow tract. The hypertrophied segment can cause obstruction when the heart rate is high or preload is low, but might not manifest symptoms otherwise. HCM must be considered in the young or adolescent patient who presents with syncope or near syncope, chest pain, or palpitations. Especially concerning features are symptoms in the setting of exertion, and a family history of unexplained or sudden cardiac death. These patients are treated with therapies to enhance cardiac filling and preload to prevent malignant arrhythmias (e.g. heart rate slowing with beta blockade, exercise avoidance, ICD).
Due to the left ventricular hypertrophy, HCM classically presents on ECG with "dagger" Q-waves in the lateral leads (deep and narrow, often < 40ms in duration) best seen in V5-V6, I, and aVL, along with large voltages across the precordium secondary to increased LV muscular mass and nonspecific ST segment and T-wave changes.

Figure: Hypertrophic cardiomyopathy. Note the dagger-like Q-waves in the lateral lead. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
Carditis
Inflammation of the myocardium can arise from both infectious causes (e.g. Coxsackie virus, myocarditis) and inflammatory sources (e.g. Kawasaki disease). Patients with carditis can present with evidence of cardiogenic shock or new heart failure. The ECG in these patients can show nonspecific signs, such as sinus tachycardia (most common), or signs classically associated with acute coronary syndromes such as ST elevations, ST depressions, or T-wave inversions. You should consider carditis when evaluating patients with possible cocaine or other stimulant ingestions.

Figure: EKG in patient with viral myocarditis. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
Prolonged QT
Abnormal repolarization of the heart can be caused by multiple genetic mutations and medications. These cause the T-wave to separate from the QRS complex, resulting in a prolonged QT interval. Patients with long QT can be asymptomatic or have a personal/family history of syncope or sudden cardiac death. Drugs such as macrolide antibiotics, ondansetron, antipsychotic agents, and phenothiazines also prolong the QT interval. If a subsequent ventricular depolarization stimulus (QRS complex) falls during a particularly sensitive part of the refractory period (R on T phenomenon), a ventricular arrhythmia known as torsades de pointes can arise.
Brugada Syndrome
This is a common cause of otherwise unexplained sudden cardiac death. Brugada Syndrome has multiple subtypes, but most are due to an abnormality in sodium channel function leading to abnormal repolarization. Like prolonged QT, it can lead to an ill-timed electrical impulse that can precipitate malignant arrhythmias, such as ventricular fibrillation or ventricular tachycardia. Brugada Syndrome classically presents as syncope or sudden cardiac death. It is characterized by three separate morphologies (types one, two, and three), with type one being most common. Type one manifests as a pseudo-RBBB appearance in the right-sided precordial leads (V1, V2), with down-sloping ST elevation leading to an inverted T-wave. Brugada Syndrome is managed with placement of implantable cardioverter-defibrillator (ICD).
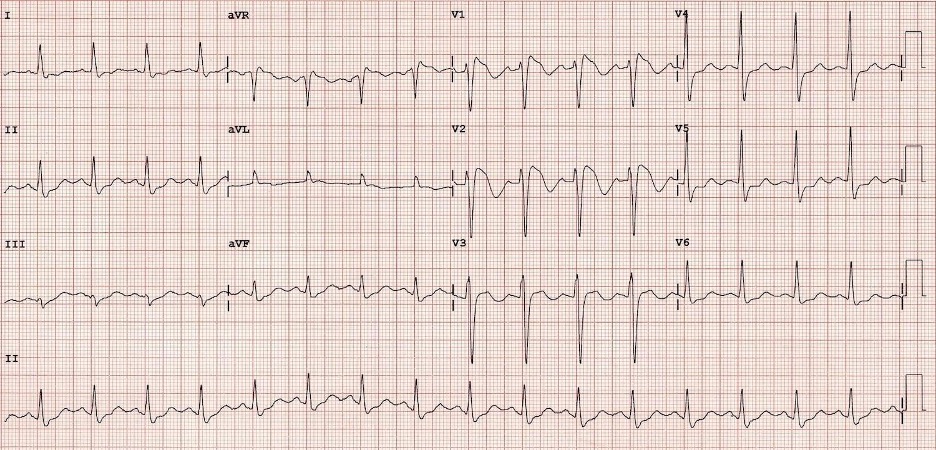
Figure: EKG from patient with Brugada Syndrome. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
Congenital Heart Diseases (CHD)
It is estimated that approximately 1% of newborns are affected by CHD, ranging from trivial or transient hemodynamic implications to potentially lethal anatomic defects. While current prenatal ultrasound and screening are highly effective at diagnosing many of the most severe CHD pathologies, an undiagnosed CHD should always be considered in an ill-appearing newborn regardless of their perinatal history or the presence of cyanosis.
While ECG is not diagnostic of any CHD, it can be a helpful tool in evaluating for cardiac pathologies. A normal ECG does not rule out CHD. There are several findings which, when found in the correct clinical context, can be suggestive of an underlying cardiac pathology and warrant consultation with pediatric cardiology.
- Left Ventricular Hypertrophy (LVH) in neonatal ECG: Due to the RV dominance described above in neonatal physiology, the findings of LVH on neonatal ECG can suggest the presence of left-sided obstructive lesions (e.g. aortic stenosis, coarctation of aorta).
- Superior Axis Deviation: An axis that is deviated to the extreme right (-90° to 180° or 180° to 270°) is frequently seen in patients with atrioventricular canal defects. Patients with Trisomy 21 (Down Syndrome) are at higher risk for this CHD.
- Normal ECG parameters for rate, axis, and intervals are age-dependent.
- Dominant R-waves in V1 can be normal from birth through three years due to transition from pre- to postnatal physiology.
- Diagnosis of cardiac pathology is rarely made by ECG alone - consider all findings in the context of hemodynamics and history.
- Supraventricular tachycardias (SVT) are the most common pathologic pediatric arrhythmia. When evaluating and treating SVT in pediatrics, keep a running rhythm strip to facilitate better understanding of underlying rhythm during conversion attempts.
After reviewing the ECG with cardiology in real-time, it was agreed that the patient is likely in a supraventricular tachycardia rhythm. While fussy, she remained alert and interactive, so an attempt at vagal maneuvers was made. A bag was filled with crushed ice and some water, then applied to the patient's face, carefully avoiding pressure on the eyes and over the airway but covering most of her cheeks and forehead. The ice compress was applied, with no change in rhythm on the ECG, and the HR remained >250 bpm. IV access was obtained, and the team decided to administer adenosine for cardioversion. A running ECG rhythm strip was started prior to rapid IV push of adenosine. Following administration of adenosine, there was a pause in heart rate, which then resumed with the following ECG.
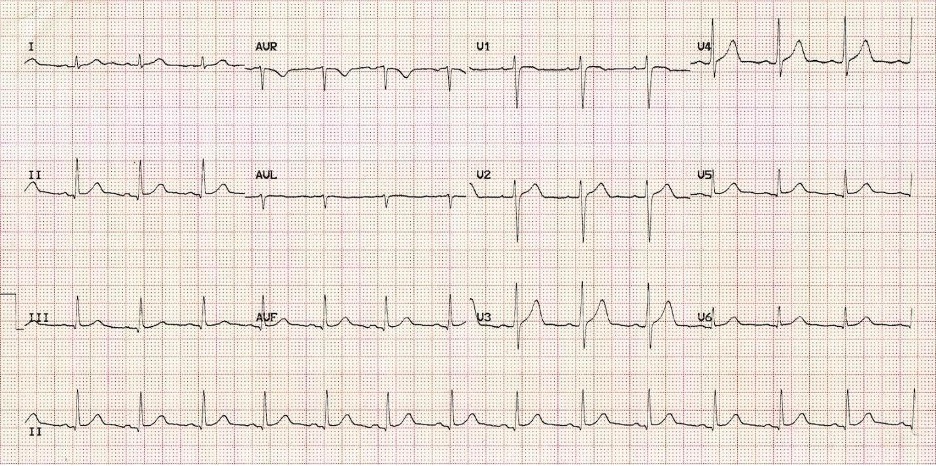
Figure: Patient's EKG after adenosine. Image courtesy of Life in the Fast Lane. Image used under the Creative Commons Attribution 4.0 International License.
You informed cardiology that the post-adenosine EKG shows a sinus rhythm with visible P-waves. The patient remained alert and interactive throughout the intervention. She was admitted to the inpatient pediatric cardiac unit for further evaluation and management of her arrhythmia.
- ECG Images. Life in the Fast Lane.
- Lambrechts L, Fourie B. How to Interpret an Electrocardiogram in Children. BJA Educ. 2020 Aug.
- O'Connor M, McDaniel N, Brady WJ. The Pediatric Electrocardiogram Part I: Age-Related Interpretation. Am J Emerg Med. 2008 Feb.
- Park MK, Guntheroth WG. How to Read Pediatric ECGs. Elsevier Health Sciences. 2006.
- Saarel EV, Granger S, et al. Electrocardiograms in Healthy North American Children in the Digital Age. Pediatric Heart Network Investigators. Circ Arrhythm Electrophysiol. 2018 Jul.
